Introduction
Inhibitors
Competitive inhibitors
Non-competitive inhibitors
Introduction
In metabolic pathways, reaction rates can be determined by the relative concentration of substrates and products of a reaction or series of reactions. The substrate presence can drive the reactions to continue for as long as there is substrate available, or alternatively the reactions could continue for as long as the product is being removed, in order to maintain a constant level.

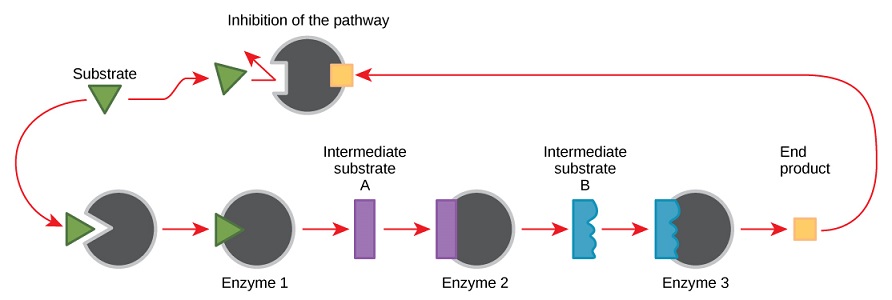
Feedback inhibition is when the presence of a threshold amount of product signals the reaction(s) to halt. This is also known as end-product inhibition.
Inhibitors
Inhibitors are molecules which interfere with the substrate binding to the active site of an enzyme, slowing down or stopping the reaction. These may be reversible or non-reversible inhibitors. The reversible inhibitors can be competitive or non-competitive.
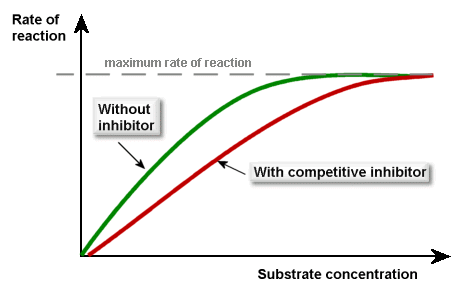
Competitive inhibitors
Competitive inhibitors have a similar 3D shape to the substrate, hence they can bind to the active site of the enzyme, preventing the substrate from doing so.

The competitive inhibitors compete (as you’d expect) with the substrate for the active sites of the enzymes. If more substrate is added, then the inhibitors’ effect will be diminished. This is what the graph looks like (make sure you can recall this):