Membrane Proteins
Transport
Diffusion
Osmosis
Facilitated Diffusion
Active Transport
Signal Transduction
Chemical
Electrical
Action Potential
The Myelin sheath
Synapse
Introduction
Since membrane proteins are found at the interface between cells, unsurprisingly one of their key functions is transport of molecules and ions across the membrane, since many can’t make their way on their own.
The main properties of molecules that determine how they may be transported across a membrane are solubility, size and charge.
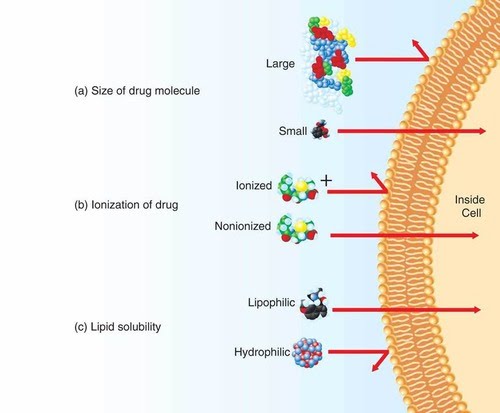
Transport
Diffusion
Diffusion = the spread of particles from a region of higher concentration to a region of lower concentration, until the particles are evenly spread out.
Diffusion takes place when you use a spray in a room, for example. The particles in the spray move randomly, knocking each other, which results in them spreading throughout the room gradually, from high concentration to low concentration. Therefore, diffusion acts down (or along) a concentration gradient.
It is important to know what affects the rate of diffusion. These are:
-
-
- Surface area – The greater the surface area, the faster diffusion will occur
- Difference in concentration –The higher the difference (the steeper the gradient), the faster diffusion will take place.
- The thickness of the exchange surface – The thicker the exchange surface, the slower the rate of diffusion.
-
Of course there are other factors such as temperature (increased kinetic energy results in faster diffusion) and the diffusion pathway (distance). The latter is a side effect of (3.) The thickness of the exchange surface, in some respects.
Osmosis
Osmosis is the diffusion of water across a semi-permeable membrane. The “concentration” of water is referred to as water potential. So osmosis is the ….
