Osmoregulation
The nephron
Urine analysis practicals
Endocrine functions of the kidney
Kidney disease
Advanced tissue technologies
Osmoregulation
Here’s a fancy topic of the newer spec… I’ve never done the kidney so I spent my post-A levels life in total kidney ignorance and utter lack of knowledge of my urination habits and their complexity, oh what a fool I have been. You on the other hand are going to have the damn honour of actually having a clue about how the brain and the kidneys freshen our blood up all the time. Oh ye enlightened children, rise.
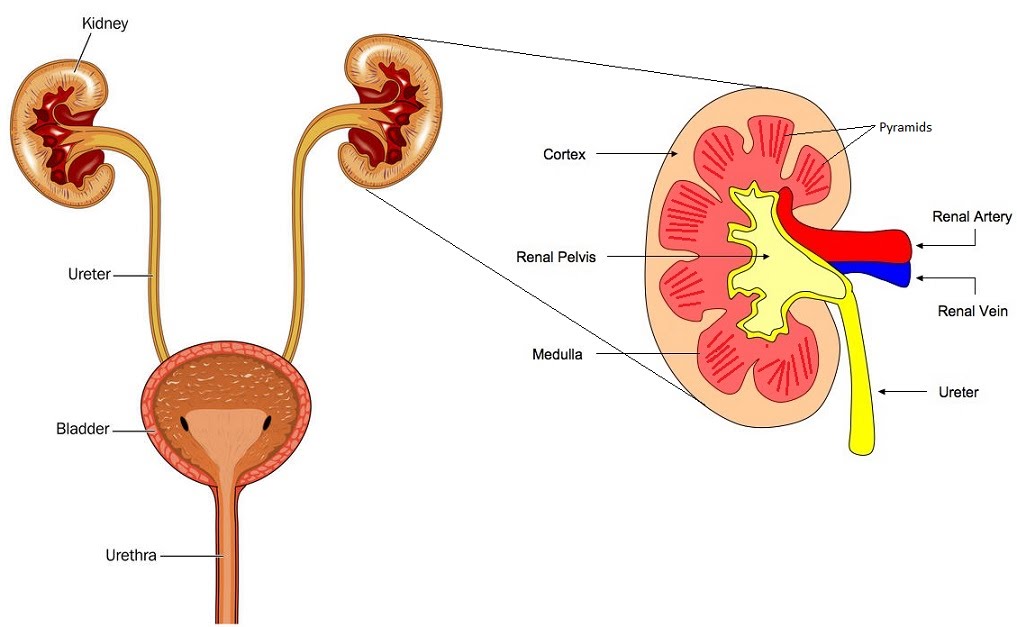
Osmoregulation refers to the control of water potential of the blood. The blood is complicated, it has all these ions and proteins and stuff. Cells use various things up all the time and some more often than others at different times, night, day, sweat, tears, etc.
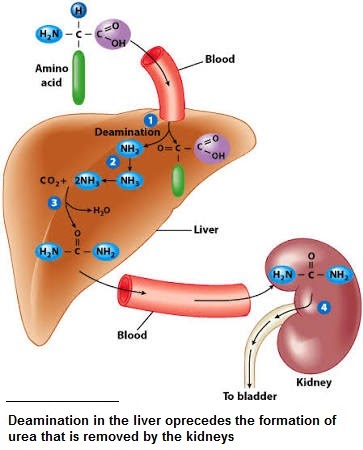
Molecules undergo deamination in the liver. This means they have an amino group removed. Since these amino group form ammonia which is toxic, it must be converted to safe urea in the blood before being excreted in urine by the kidneys. This is accomplished through the reaction with carbon dioxide.
here are systems in place that keep the blood at the right composition and pressure. The hypothalamus and posterior pituitary in the brain release a hormone into the blood that reaches the kidney and enables its cells to take up more water, to prevent it being wasted in urine as the case may be. This is detected by osmoreceptors…
