Transplantation and immunosuppression
Introduction
Kidney transplant
Donors
Blood groups
Microbial resistance
Monoclonal antibodies
Introduction
Organ transplantation between individuals, such as kidney transplants, expose the recipient to the foreign antigens of the donor. Upon identifying the foreign antigens, the recipient’s immune response mounts a defence via production of specific B- and T-cells, attacking and damaging the new organ.
Kidney transplant
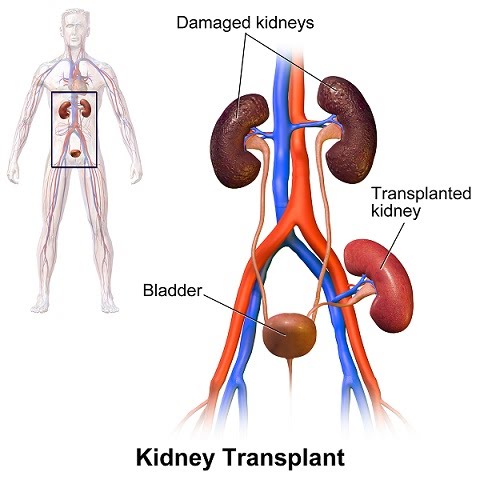
Kidney transplantation involves using a dead or live donor’s kidney to rescue kidney function of a patient on dialysis, or preemptively before they require dialysis. It is a 3-hour operation that attaches the new kidney to the patient’s blood supply, without removing the old kidneys (their removal has been shown to increase surgery outcome problems). Kidney transplants successfully increase lifespan, with better results seen the sooner it is performed relative to time on dialysis.
Donors
Donors are relatives, friends, altruistic strangers or sellers on the kidney market which is legal and supported in some parts of the world, such as Iran. Different locations have different approaches to kidney donation, with some prohibiting payment, and others openly encouraging donation through financial incentives. Successful donation chains have been established in some countries. They work by passing on incompatible kidneys from donors aimed at their relatives, onto strangers who are compatible with their kidney. In exchange, they get a compatible kidney from another stranger, and so on and so forth, creating a donation chain that addresses compatibility issues while encouraging kidney donation and extending the available organ pool.
Patients reach healthy kidney function following the transplant within a few days to weeks, and must adhere to a strict immunosuppressant drugs course indefinitely to maximise the success of their transplant.
Immunosuppressant drugs act by preventing the production of specific B- and T-cells. They work by inhibiting DNA replication. Alternatively, disabling their production can be accomplished via X-ray irradiation. The patient will no longer mount an immune response via specific B- and T-cells when presented with the antigens of the new organ, but as a side effect, will be….

