Metabolic pathways
Membranes
Enzymes
Inhibitors
Substrate and enzyme concentration
Metabolic pathways
Right, all the reactions that happen in a body. No easy way of putting this. Voila!
It’s OK, don’t panic. Metabolic pathways are the routes from chemical to chemical that represent their various reactions inside cells, catalysed by enzymes. Enzymes are proteins that enable a much faster reaction – catalysts. These pathways cover the making and breaking of all compounds. They cover the energy-producing processes that use oxygen and glucose to make ATP (adenosine triphosphate, the cellular energy currency), the breaking down of sugars, fats and proteins from food, the building of new enzymes, the constant breaking down of waste products and creation of maintenance molecules for energy and the many functions in the body, etc.
These metabolic pathways are integrated and controlled so that connections exists between pathways, with checkpoints that can be self-regulating to ensure the right rate of reaction is taking place. Some simpler chemicals are recycled in some pathways, while others are introduced to the system from the outside e.g. through food and drink, as well as removed from they system e.g. excretion.
The pathways which use up energy to carry out their reactions are called anabolic, while those that release energy are catabolic. Anabolic pathways are biosynthetic as they assemble larger molecules from smaller units, hence requiring energy. Catabolic pathways break down larger molecules into their constituent building blocks, releasing energy. Chemical bonds contain energy, so breaking them releases it, while creating them requires it.
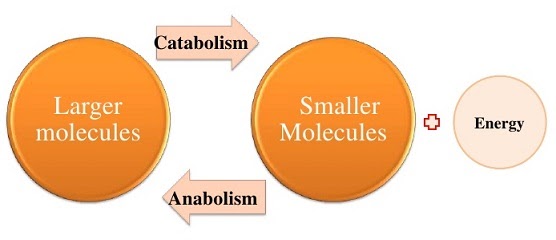
These pathways can have reversible steps where a reaction can go backwards or forwards to break down or assemble a chemical again, or irreversible steps where the broken down compound, or the newly synthesised molecule, cannot be undone and will have to follow..
