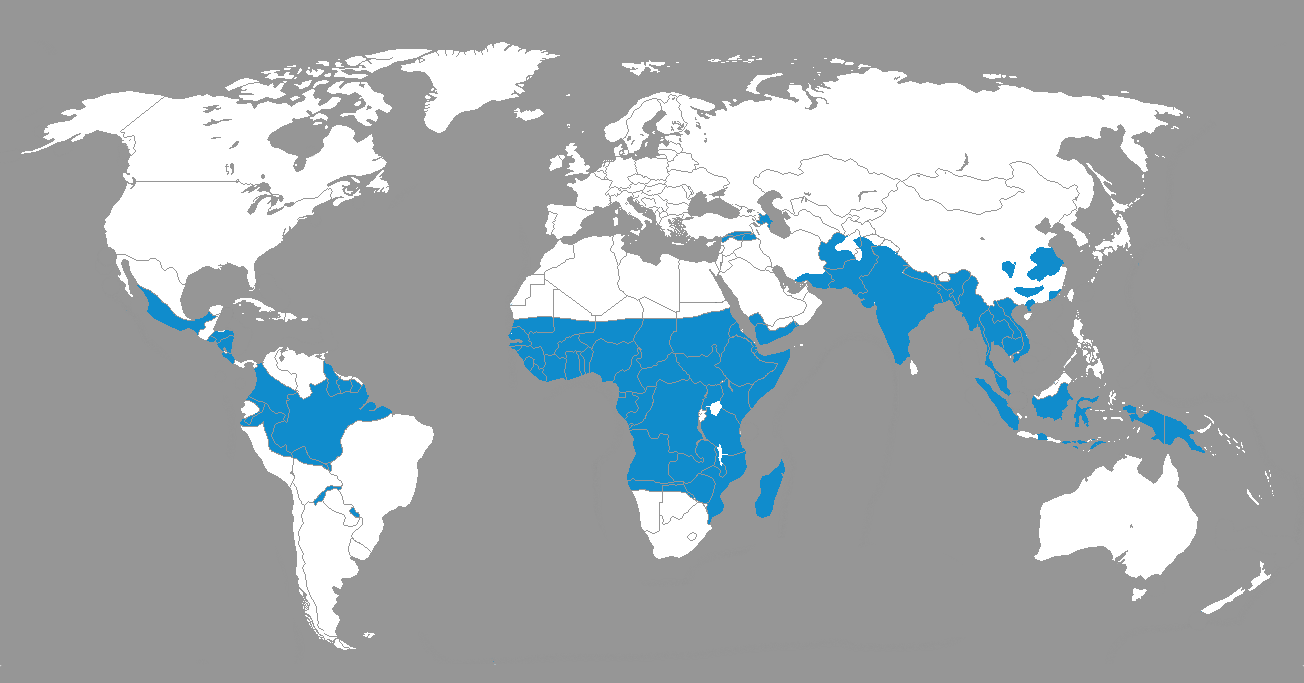
The difference between endemic and epidemic
Ethical Issues
Economically
Economically speaking, it may be more worthwhile to prevent the disease than to treat it. This is feasible in areas like China where the money needed by the affected Chinese provinces to execute this is a small percentage of the healthcare budget, but not necessarily in other parts of the world affected by malaria, such as Tanzania, where these measures would by comparison be equivalent to a large portion of the budget (a fifth)…