Actin
What happens to the I band during contraction?
Actin
During contraction, the myosin doesn’t actually get shorter. It is the actin that slides over to decrease the overall length of the sarcomere.
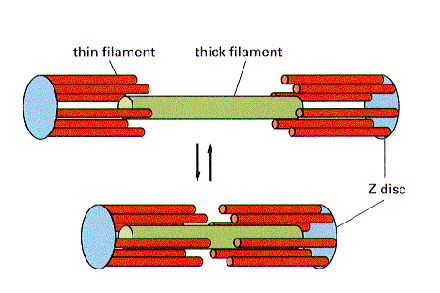
This is a really good diagram for understanding the basic principle of actin (thin filaments) sliding over the myosin (thick filaments) to shorten the sarcomere and achieve contraction. If you understand this, you won’t be shocked by the inevitable trippy questions that examiners will hurl at you in the exam about fancy H/I/A bands/lines/zones. These randomly named lines/zones/bits/pieces are totally arbitrary. If you must know, the A band and I band referring to the length covered by myosin and actin respectively were established as a result of how they appear under a microscope.
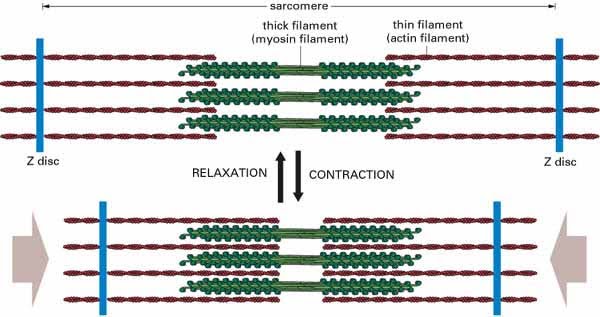
Another diagram for the visually-inclined.
Let’s go back to…
