Parasite niches
Transmission and virulence
The immune response
Parasite life cycles
Challenges in treatment and control
Parasite niches
Parasites are symbiotic species that rely on their host for nutrients, like predators rely on prey. This is at the expense of the host, which often suffers illness as a result, or a damaged reproductive function. Unlike predators that have a lower reproductive capacity compared to their prey, parasites have a higher reproductive potential than their host.
An ecological niche is the set of tolerances and requirements a species has in its immediate living space. For example, birds make use of a tree’s branches to house nests, while fungi make use of a tree’s trunk to grow. The same habitat can have many different niches that can be populated by different species.
Parasites have very narrow niches because they are specific to their host. Human head lice cannot survive anywhere except the human scalp. This also means that the molecular processes that parasites rely on in their host are very similar and overlap with those of the host – one reason developing effective treatments against them is challenging. This interdependence means that some processes carried out by the host on behalf of its parasite renders the parasite degenerate i.e. unable to fulfil them by itself, and lacking in structures and organs other species might have.
The aforementioned head lice are an example of an ectoparasite, parasites that reside on the surface of their host. Internal parasite such as tapeworm are called endoparasites.
Tapeworms reside in the small intestine where they can conveniently tap into the host’s nutrients, and so interfere with the normal absorption of the host’s nutrients into its bloodstream, thereby depriving the host to potentially dangerous levels of malnutrition and other side effects such as anaemia and fatigue.
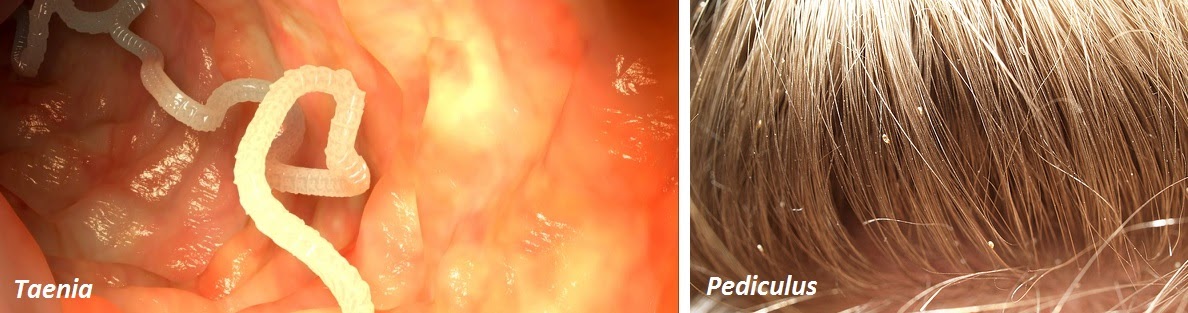
Lice live on the body, such as pubic lice or on eyelashes, or the scalp in the case of head lice, and feed on the host’s blood. Sensitivity to lice saliva causes itchiness, and some lice can be vectors (carriers) of dangerous infectious agents such as Epidemic typhus and Trench fever.
A vector species may act as a bridge between the parasite and its host by transmitting it to the host where the parasite reaches sexual maturity – its definitive host. The vector or another species may also act as an intermediate host for a required precursor stage of the parasite’s life cycle…
