Introduction
Plasma Membranes
The fatty acids can be simplified in drawing
Ester bonds
Emulsion test
Introduction
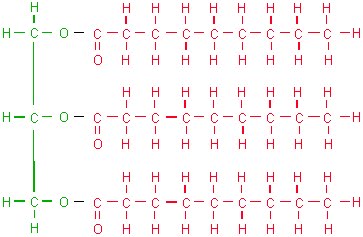
Membranes are made of phospholipids, which are made of lipids. Lipids are the stuff of oils, fats and waxes. Unlike proteins and carbohydrates, lipids are not polymers. Lipids which store energy are triglycerides, while those which form membranes are phospholipids. Triglycerides are formed by a molecule of glycerol with three fatty acids attached. The reaction which results in triglycerides is condensation.
Glycerol (green) + 3x fatty acids (red)
The fatty acids can be simplified in drawing:
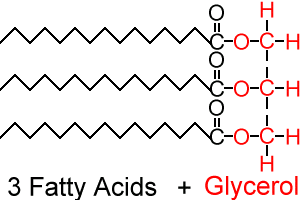
The bonds formed (C-O) are called ester bonds. Fatty acids can be saturated or unsaturated (monounsaturated; polyunsaturated). Saturated fatty acids have all their carbon (C) atoms linked to hydrogen (H) atoms, hence saturated with hydrogen. If there is a carbon atom with a double bond to its neighbour carbon atom, then it will only have one bond to a hydrogen atom, hence it is unsaturated. If there is one double bond present, the fatty acid is monounsaturated. If there are multiple double bonds present, the fatty acid is…