The cytoskeleton
Cell cycle and mitosis
Cell division regulation and apoptosis
Stem cell differentiation
The cytoskeleton
Cell division requires the active remodelling of the cell’s cytoskeleton. The cytoskeleton is made of multiple kinds of protein that offer it the right structure and aid during the process of division.
The centriole is a tubey spaghetti thing that aids in cell division when the duplicated chromosomes need to move into their subsequent new offspring cells from the parent cell (during mitosis).
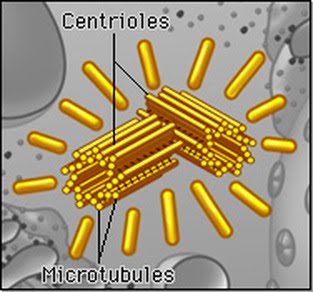
They’re made of a special protein called tubulin because they’re tubeeeeeeeeeeeees. Why didn’t they call it spaghettulin? I guess spaghetti aren’t hollow but…
Microtubules also play a key part in how cell organelles are moved and placed within the cell. Two associated centrioles form the centrosome. The microtubules are also where spindle fibres extend from when they separate chromosomes during cell division.
-Cell cycle
The cell cycle refers to the distinct stages through which a cell goes, from the moment it becomes a cell to the moment it divides to result in 2 separate cells. Bear in mind that some cells cease to divide any longer after a certain period of time, depending on cell type. If that’s the case, they are said to be in resting phase termed G0.
A decrease in the cell cycle beyond appropriate levels would lead to degenerative disease, while too high an increase would cause tumours. Therefore, a balanced cell cycle is key to the good functioning of the body.
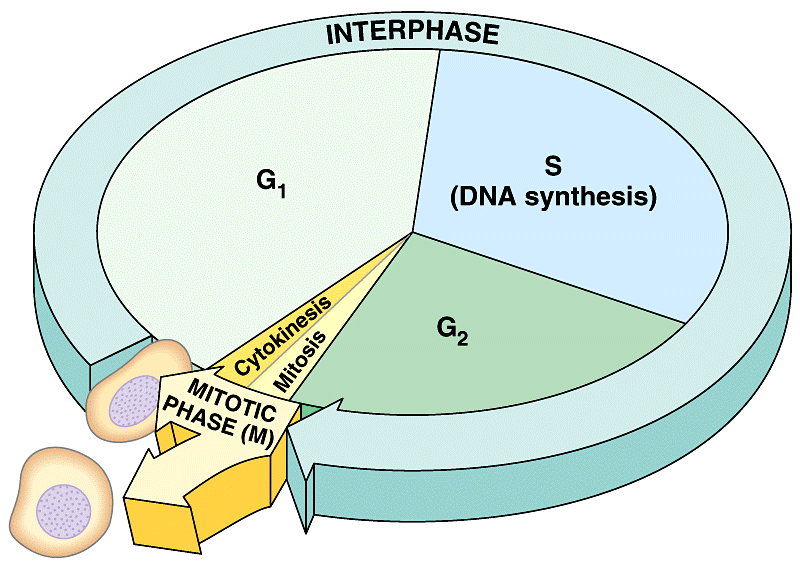
Within the dividing cell, it starts with gap 1, G1, continues into the S phase (S is for Synthesis) where DNA replicates, followed by gap 2, G2, and ending with mitosis.
G1 and G2 may sound like codes for some complex enzymes, but they are mere notations for gaps 1 and 2, which are just that: gaps between mitosis and DNA replication (in the S phase) respectively. G1 through to G2 – that’s G1, S phase and G2 – are all stages which collectively are known as interphase. Inter = between; phase = …phase, so interphase is just the stage between a cell’s creation and that cell’s division by mitosis.
Interphase is by far the stage in which most cells are in most of the time. The other stage, the small one, is called the mitotic phase and it encompasses mitosis (prophase, metaphase, anaphase and telophase) plus cytokinesis.
-Overview of Mitosis
Mitosis is the process by which cells divide to achieve growth and repair by simply increasing cell number. For unicellular organisms, cell division is actually their reproduction itself (asexual reproduction). The dividing cell is called the parent cell, and the resulting two cells have inappropriately been called daughter cells by scientists so far. Now because cells don’t have a damn gender and we are better than accepting silly nonsensical received wisdom about what to call these cells, we will call them offspring cells instead. The offspring cells are genetically identical i.e. clones, as they contain copies of the parent cell’s DNA.
-Stages of Mitosis
Prophase, metaphase, anaphase, telophase and….
