Meiosis
Antenatal care
Assessing foetal development
Meiosis
Meiosis is a type of cell division which results in 4 cells that are genetically non-identical from one parent cell. In order for once cell to divide to result in 4 cells, how many divisions must take place?
Two. 1 cell becomes 2, then 2 become 4:
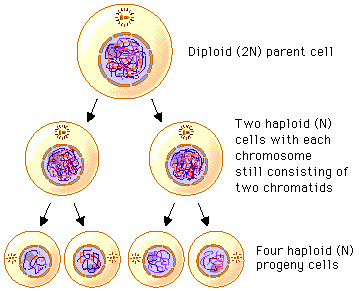
The first division is called meiosis I, and the second is called meiosis II.
…so far so easy? (it should be!)
Cells resulting from meiosis are gametes such as egg cells and sperm cells, hence meiosis only occurs in sexually reproducing organisms. There are 2 key points about this:
1.Gametes are genetically unlike one another – while cells in other tissues such as muscle or blood must be genetically identical to one another (clones), the very basis of sexual reproduction is genetic diversity. So somewhere in the process of division, something takes place which creates genetic diversity (we’ll come to that shortly).
2. If gametes are to fuse and result in a new organism, it is essential that the number of chromosomes should stay constant. Humans have 46 chromosomes in each cell (of course, apart from cells without DNA in them, and “spoiler alert!”, gametes) – if each gamete had 46 chromosomes, then fusing 2 together would result in a zygote with 92 chromosomes, whose offspring would have 184 chromosomes…
