DNA structure
What are the strands made of?
What is the centre made of?
DNA organisation
DNA structure
DNA (deoxyribonucleic acid) is a large molecule which carries the genetic information, or blueprint, of all life on Earth. Mutations arising in the DNA code account for the diversity upon which evolution by natural selection can work. Therefore, it is not far-fetched to say that DNA is one of the central, most important molecules in living organisms.
For such an important molecule, it sure looks beautiful:

DNA is a double helix i.e. two individual strands running along each other in an anti-parallel way, connected to one another by relatively weak hydrogen bonds. DNA’s structure can be learned easily by thinking about the strands and the “stuff in-between” separately.
What are the strands made of?
The strands are made of repeating units consisting of a deoxyribose (sugar) molecule with a phosphate molecule attached to it; hence, it is called a sugar-phosphate backbone.
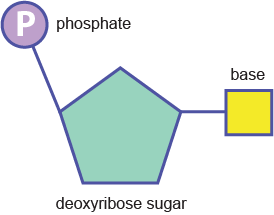
Phosphodiester bonds between nucleotides (above) create the backbone:
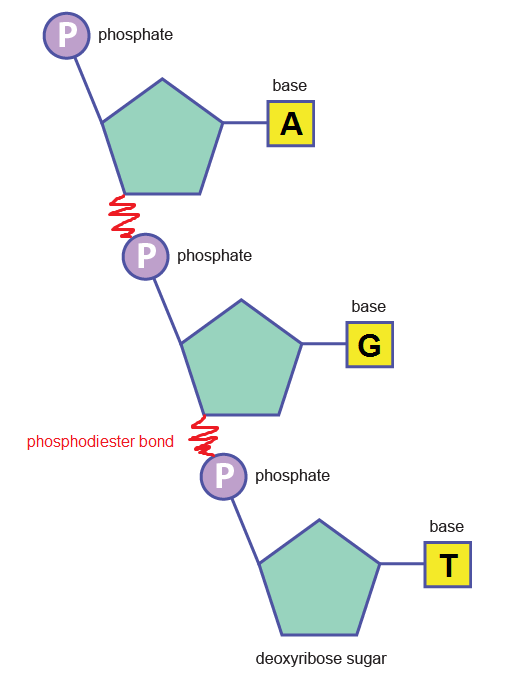
DNA is made of 2 of these strands running in an anti-parallel structure. Because the strands have a specific orientation, they are termed differently. It happens that at the end of each strand, the nucleotide with the unattached phosphate group is found at one end, while the deoxyribose sugar is found at the opposite end, with its phosphate group linked to the above nucleotide via the phosphodiester bond….
