Cell transport mechanisms
The plasma membrane
Passive transport
Diffusion
Facilitated diffusion
Osmosis
Endocytosis and exocytosis
Active transport
ATP and ADP
The plasma membrane
Armed with this knowledge of lipids, as well as carbohydrates and proteins, we can now explore the structure of plasma membranes, specifically in the context of the fluid-mosaic model. Phospholipids have a hydrophilic (water loving) head, and hydrophobic (water repelling) tails. This results in the formation of a phospholipid bilayer (double layer), which forms the basis for the plasma membrane.
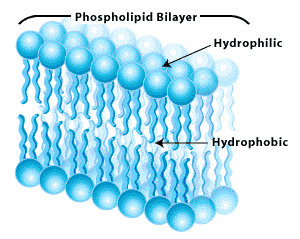
The name of fluid-mosaic model comes from:
- Fluid = the arrangement of proteins contained in the membrane is always changing
- Mosaic = the proteins present are spread around in a mosaic-like fashion.
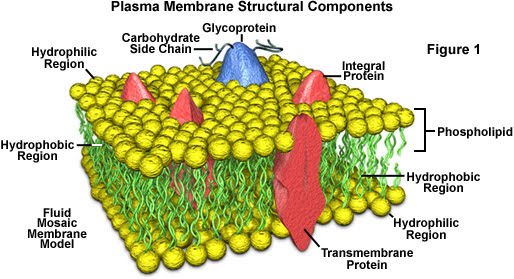
It’s pretty isn’t it? The proteins are crucial to cell communication as well as the selective permeability of the membrane. The glycoproteins’ (sugars/carbohydrates attached to a protein) side chains act as receptors. Lipid soluble stuff such as vitamins A, D and K, as well as oxygen and carbon dioxide, can pass freely though the membrane. Cholesterol can be part of the membrane to restrict the movement of other components.
The main properties of molecules that determine how they may be transported across a membrane are solubility, size and charge.
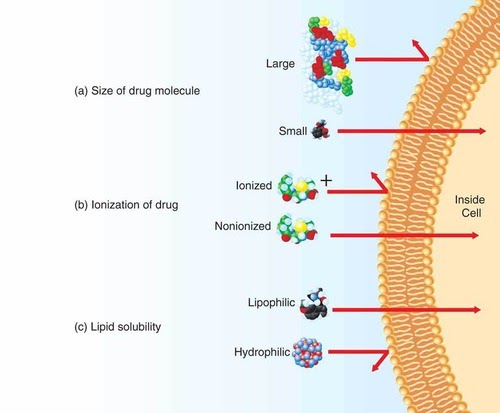
Large molecules can’t cross the membrane, charged molecules also can’t, and naturally, lipid-repelling (or water-attracting) molecules can’t. Conversely, small molecules can cross the membrane barrier, alongside molecules with no charge (nonionised) as well as lipophilic (hydrophobic) molecules.
It’s important to understand the role of microvilli. These are elongations of plasma membrane which increase the surface area available for reaction or absorption…



